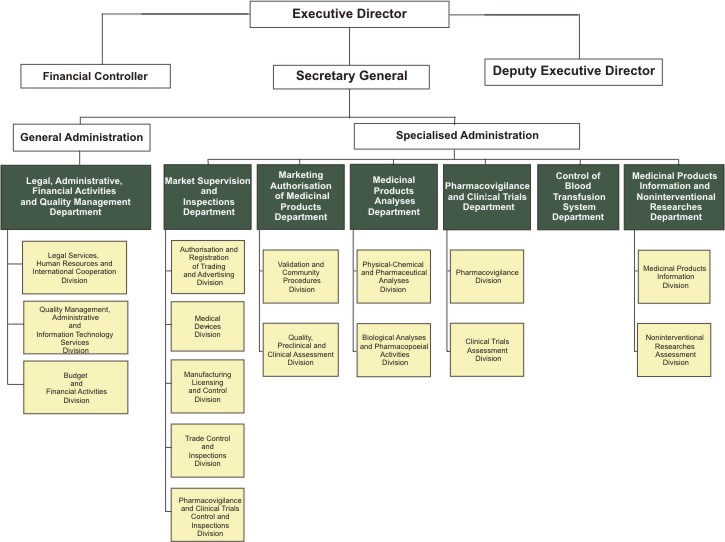
State institutions of the republic of Bulgaria
|
Parliament |
|
|
Council of Ministers Administrative registry at the Council of Ministers |
|
|
Ministry of Health |
European institutions
|
Gateway to the European Union (The official site of the European Union) |
|
|
European Commission |
|
|
European Medicines Agency (EMA) |
|
|
European Directorate for the Quality of Medicines and Healthcare, Strasbourg |
European medicines regulatory agencies
|
Austria |
Bundesamt für Sicherheit im Gesundheitswesen |
|
|
Belgium |
Directoraat generaal Geneesmiddelen Direction générale Médicaments |
|
|
Bulgaria |
Institute for Control of Veterinary Medicinal Preparations |
This email address is being protected from spambots. You need JavaScript enabled to view it. |
|
United Kingdom (H) |
Medicines and Healthcare products Regulatory Agency |
|
|
United Kingdom (V) |
Veterinary Medicines Directorate |
|
|
Germany (H) |
Bundesministerium für Gesundheit und Soziale Sicherung Bundesinstitut für Arzneimittel und Medizinprodukte Paul-Ehrlich-Institut |
|
|
Germany (V) |
Bundesministerium für Verbrauerschutz, Ernahrung und Landwirtschaft Bundesministerium für Verbraucherschutz und Lebensmittelsicherheit (Veterinary medicinal products) |
|
|
Germany (H+V) |
Zentralstelle der Länder für Gesundheitsschutz bei Arzneimitteln und Medizinprodukten |
|
|
Greece |
National Organisation for Medicines |
|
|
Denmark |
Lægemiddelstyrelsen |
|
|
Estonia |
State Agency of Medicines |
|
|
Ireland |
Irish Medicines Board |
|
|
Iceland |
Lyfjastofnun |
|
|
Spain |
Agencia española del medicamento |
|
|
Italy |
Ministero della Salute |
|
|
Cyprus |
Ministry of Health Ministry of Agriculture |
|
|
Latvia |
Food and Veterinary Service |
|
|
Lithuania |
State Medicines Control Agency State Food and Veterinary Service |
|
|
Lichtenstein |
Liechtensteinische Landesverwaltung Amt für Lebensmittelkontrolle und Veterinärwesen Kontrollstelle für Arzneimittel |
|
|
Luxembourg |
Ministère de la Santé Division de la Pharmacie et des Médicaments |
|
|
Malta |
Medicines Authority |
|
|
Norway |
Statens Legemiddelverk |
|
|
Poland |
Office for Medicinal Products |
|
|
Portugal |
Instituto Nacional da Farmácia e do Medicamento |
|
|
Romania |
National Medicines Agency National Sanitary Veterinary Agency |
|
|
Slovak Republic |
State Institute for Drug Control Institute for State Control of Veterinary Biologicals and Medicaments |
|
|
Slovenia |
Agency of the Republic of Slovenia for Medicinal Products and Medical Devices |
|
|
Hungary |
National Institute of Pharmacy Institute for Veterinary Medicinal Products |
|
|
Finland |
Lääkelaitos |
|
|
France (H) |
Agence Française de Sécurité Sanitaire des Produits de Santé |
|
|
France (V) |
Agence Française de Sécurité Sanitaire des Aliments |
|
|
Netherlands |
Staatstoezicht op de volksgezondheid Inspectie voor de Gezondheidszorg |
|
|
Netherlands |
College ter Beoordeling van Geneesmiddelen (CBG) |
|
|
Czech Repbulic |
State Institut for Drug Control Institute for the State Control of Veterinary Biologicals and Medicaments |
|
|
Sweden |
Läkemedelsverket |